Breast cancer has become the most common cancer. Statistics from Siriraj Cancer Center in 2008 showed 1,129 new breast cancers, making up 25.79% of all women’s cancers.1 Since there is no effective way to prevent breast cancer, the best that can be done is to treat the disease at its earliest stage. Breast imaging which includes mammography (MM), ultrasound (US), magnetic resonance imaging (MRI) and radionuclide scintigraphy (RNS), has proven to be the most appropriate method to detect lesions.2-5 Concerning the cost effectiveness and availability, MM and US are the studies of choice in our country.6-8
Statistics of breast tissue composition in 191,169 mammograms performed from 1995-2009 in Siriraj Breast Centre show almost entirely fat in 5.64%, scattered density in 20.36%, heterogeneously dense in 63.18% and extremely dense in 10.82%, therefore, 75% of Thai breasts can be classed as difficult breasts, in which lesions may be completely obscured.9 Mammography has limitations in detection and evaluation of breast lesions in mammographically dense breasts, palpable mass in young women, pregnant and lactating breasts, acute inflammation, post-operation, trauma, male gynecomastia and assessment of augmented breasts. US has no such limitation. The masses in MM are notified either as round, oval, lobular, which are less significant or as irregular, microlobular and speculated, which are more suspicion. There might be associated findings, such as the presence of microcalcifications and architectural distortion.10,11
US is much more informative in evaluation of breast mass, for its shape (round, oval or irregular), orientation (parallel, non-parallel), margin (circumscribed or not, sub grouped into indistinct, angular, microlobulated or speculated), lesion boundary (abrupt interface or echogenic halo), echo pattern (anechoic, hypoechoic, isoechoic, hyperechoic and complex), posterior acoustic features (no posterior acoustic features, enhancement, shadowing and combined pattern), surrounding tissue (in ducts, straightening or thickening of Cooper’s ligament, edema, skin thickening, architectural distortion), vasculature and other specific findings. Therefore, apart from seeing the mass, US can define the mass as:
The management of each subgroup is different
We compared results between mammography, pathology and US results. The outcome of using both studies together yielded a much more impressive results, namely that when the mass looked benign, it was true benign in 94.9%, probably benign was benign in 88.57%, indeterminate is 50 +/- 3.49% and malignant was true malignant in 95.78% of cases.12-15
Of 26,741 MM with US performed during 1999–2004 in Siriraj Breast Centre, US was able to detect 141 cancers in MM negative (182 US BIRADS 4, 32 US BIRADS 5). Concerning detection of breast mass, there were 13,109 cases that in both MM & US were positive, 14,220 cases that were positive in US, negative in MM.16
US of axillary adenopathy is superb, giving more internal architecture details, pathology and including more nodes than can be seen by MM. Fine-Needle Aspiration (FNA) of the abnormal node can be performed in one setting, which is far better than to search and do excisional biopsy of sentinel lymph node, performed in the operating room. Normal nodes were also studied.17
US has limitation in detection of microcalcifications, especially those with no associated mass. Of 39,830 screenings in Siriraj Breast Centre in 2004, there were 84 breast cancers. We found 19 cases (22.62%) with microcalcifications alone, which were negative in US, and 106 cases (6.02%) of 1761 cancers in 22,442 diagnostic cases had microcalcifications alone.18 Those are the cases that will be missed if US was performed without MM. However, if the lesion is extensive, US may show where the lesion is and US guided core needle biopsy (CNB) can be performed, with proven specimen radiograph to show the presence of the microcalcifications.
US has technical drawbacks in limitation of field of view (FOV): it is not reproducible, time consuming, requires high skill/experience, operator dependent, not fit for screening. These problems can be overcome by using an automated breast US volume scan, which is a quick study that provides multiplanar images with very high resolution. It is reproducible, thus very beneficial to be used in a comparison study. The automated breast volume scanner (ABVS), like computed tomography (CT) volume scan provide automated acquisition of a large number of thin two-dimensional (2D) slices to produce a threedimensional (3D) volumetric data set. A reconstruction by post-processing is instantly performed in multiplanar: sagittal, coronal, radial, anti-radial, cranial-caudal (CC), Mediolateral-oblique (MLO), multislices: thin or thick slices, 3D navigation, tomographic display 3D which allows for correlation with mammography or MRI.19,20
In 2010, we did a study on Correlation of ABVS and HHUS after mammography in 212 female patients at Breast Center of Wattanosoth Hospital. Their average age was 48.06 years. HHUS was interpreted and reported by radiologists immediately. ABVS was interpreted and recorded by one radiologist 3 months later. The results of both studies were recorded, analyzed and correlated.
There were 504 lesions, interpreted as BIRADS 1 in 37 cases, BIRADS 2 (simple cyst) in 77 cases, BIRADS 3 in 63 cases, BIRADS 4 in 31 cases and BIRADS 5 in 4 cases. Both studies agreed in 306 lesions, there was agreement and ABVS showed additional lesions in 13 cases, the studies were not in agreement about 35 lesions, ABVS showed negative but HHUS showed positive in 24 lesions, HHUS showed negative but ABVS showed positive in 104 lesions and there were pitfalls in ABVS results in 24 lesions. (Table 1)

Table 1: Correlation of ABVS and HHUS findings
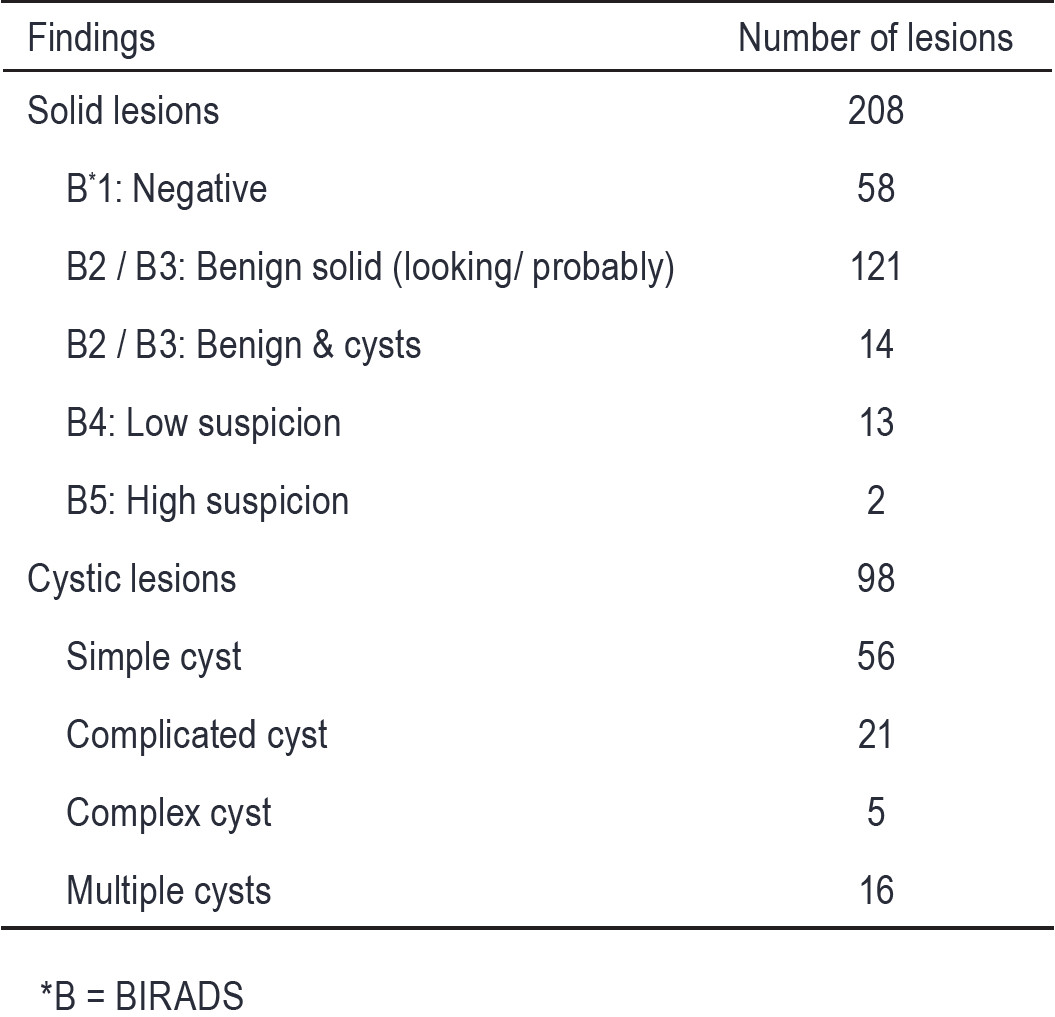
Table 2: Correlation of 306 agreed findings by ABVS and HHUS.

Table 3: The ABVS and HHUS agree findings but ABVS shows 13 more lesions.
Details of the findings in each category are as follows:

Figure 1: MM does not show any lesion. HHUS shows a microlobulated solid mass, proven to be invasive ductal carcinoma (IDCA). ABVS shows the same finding in multiple images at 2 mm thickness and multiplanar, including axial, sagittal and coronal planes.
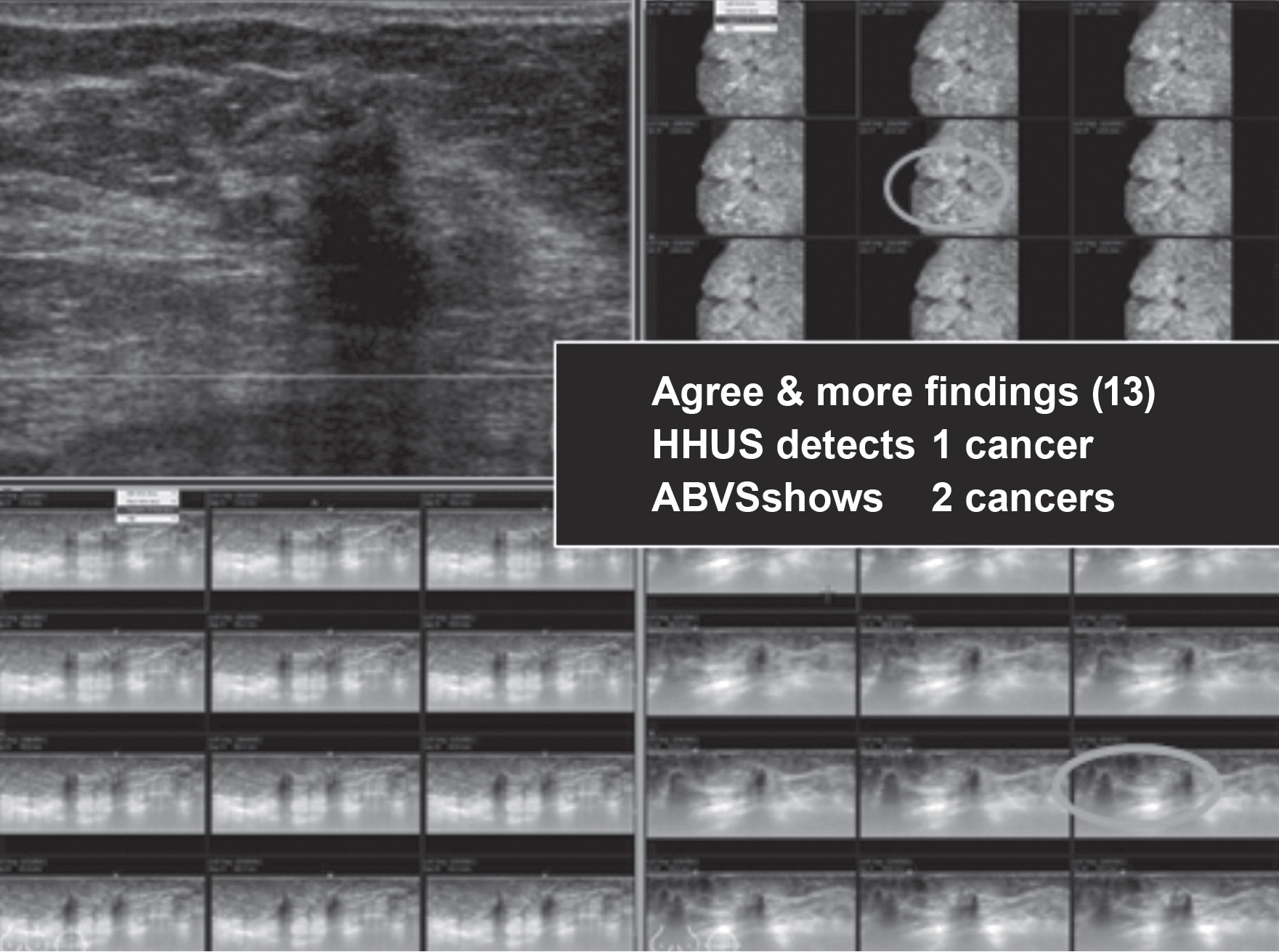
Figure 2: HHUS shows a small solid mass with increased depth to width ratio and strong acoustic shadowing, proven to be invasive ductal carcinoma. ABVS shows two nearby lesions of the same findings, displayed in multiple images and multiplanar, including sagittal and coronal planes.
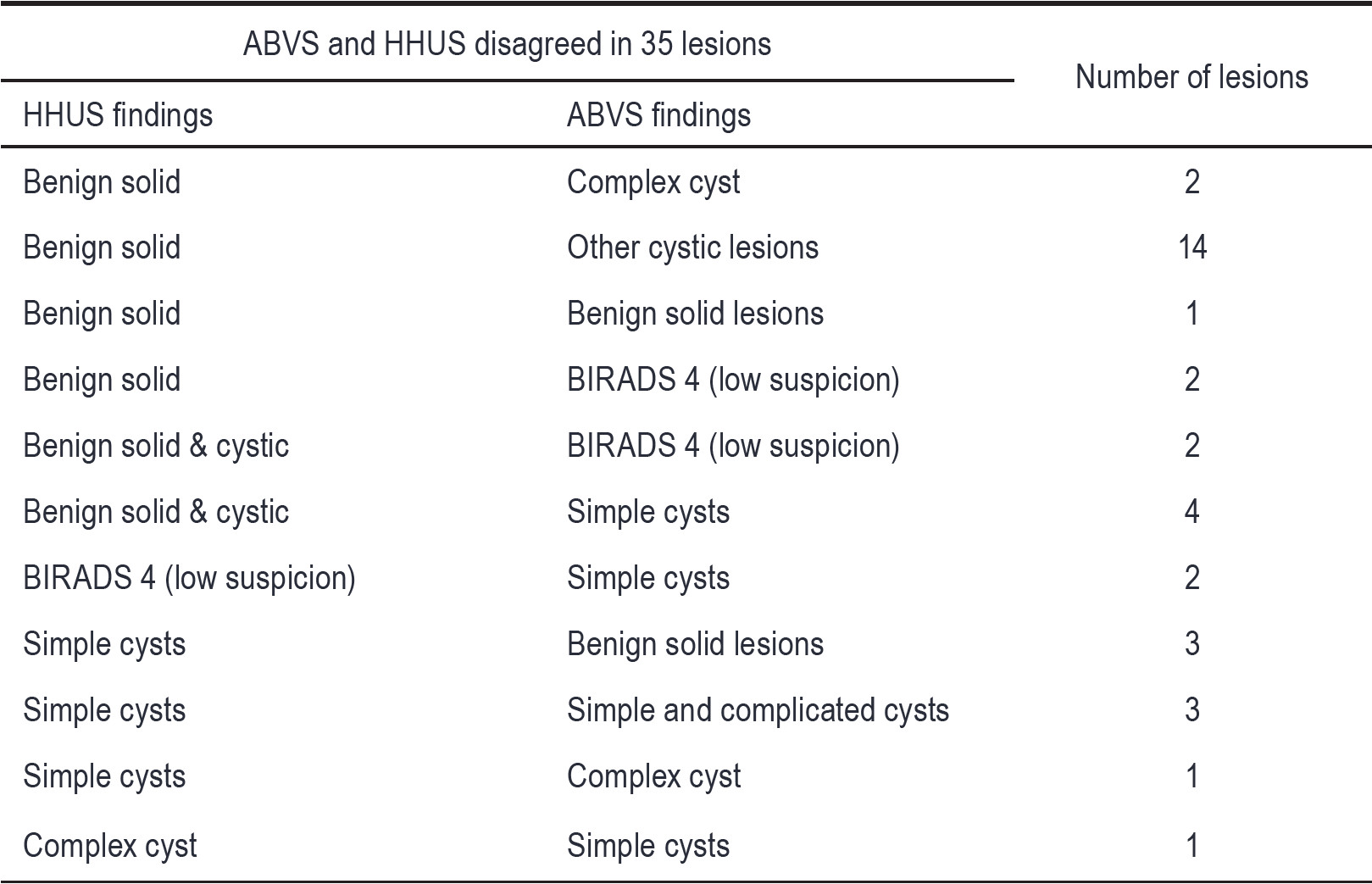
Table 4: The ABVS and HHUS disagreed in 35 lesions.
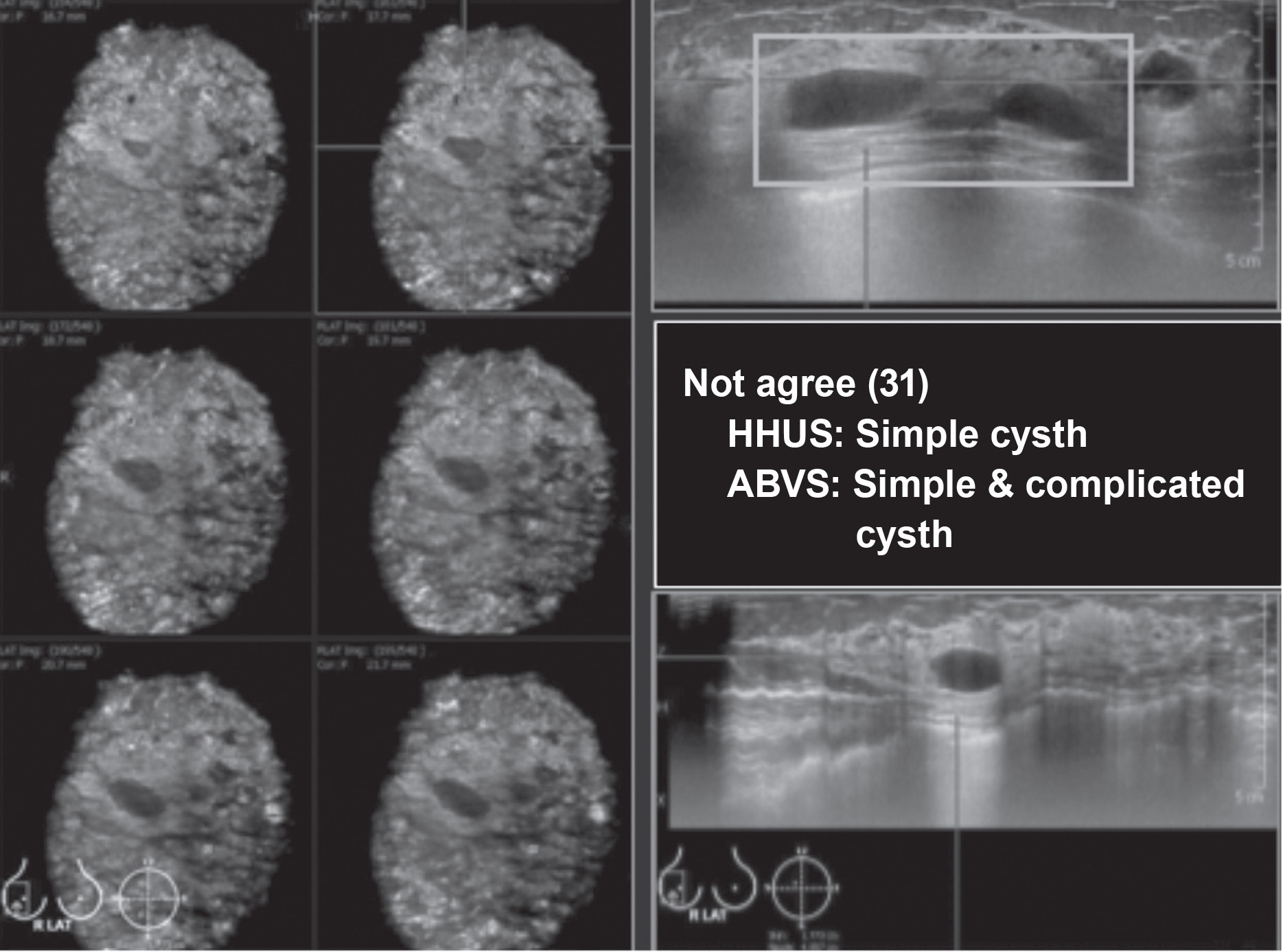
Figure 3: HHUS shows multiple anechoic simple cysts. ABVS shows some cysts with low echoic contents, compatible with complicated cyst. The large FOV allows good comparison of the lesions in one image, displayed in multiple images and multiplanar, including axial, sagittal and coronal planes.
From our study ABVS showed much more benefit over HHUS. However, ABVS had drawbacks, for example:
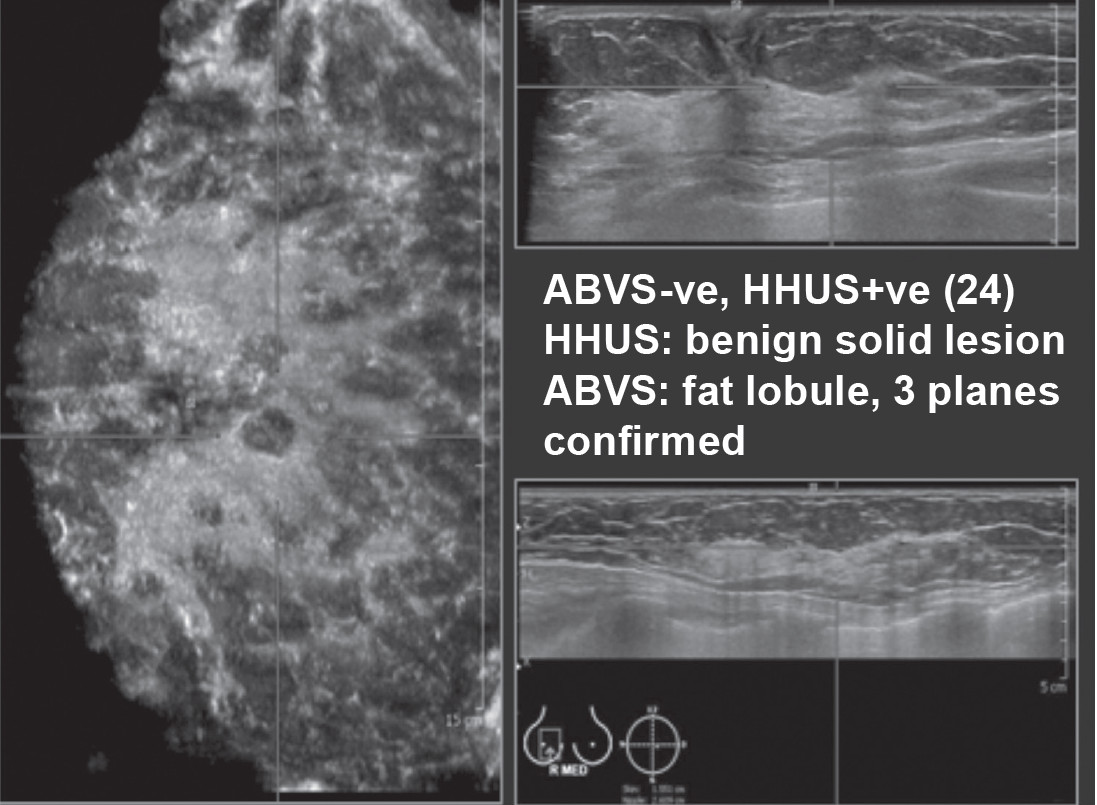
Figure 4: HHUS shows hypoechoic lesions, while this case, ABVS shows the low echoic lesions continues with subcutaneous fat in another 1 – 2 views, compatible with fat lobule and excluded these false positive lesions seen by HHUS.
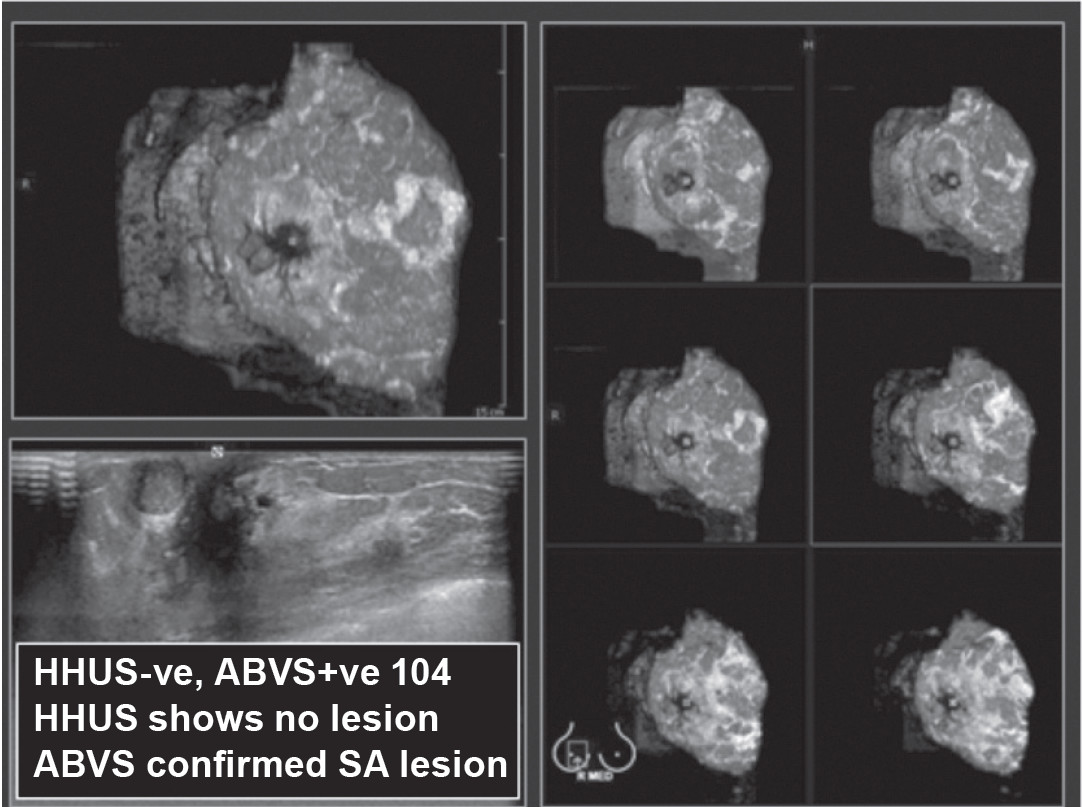
Figure 5: HHUS detects no lesion. ABVS clearly demonstrates a lobulated hypoechoic solid mass occupied almost entirely inside a cystic lesion, compatible with a complex cystic lesion, confirmed to be a papilloma by core needle biopsy (CNB) and surgical specimen.
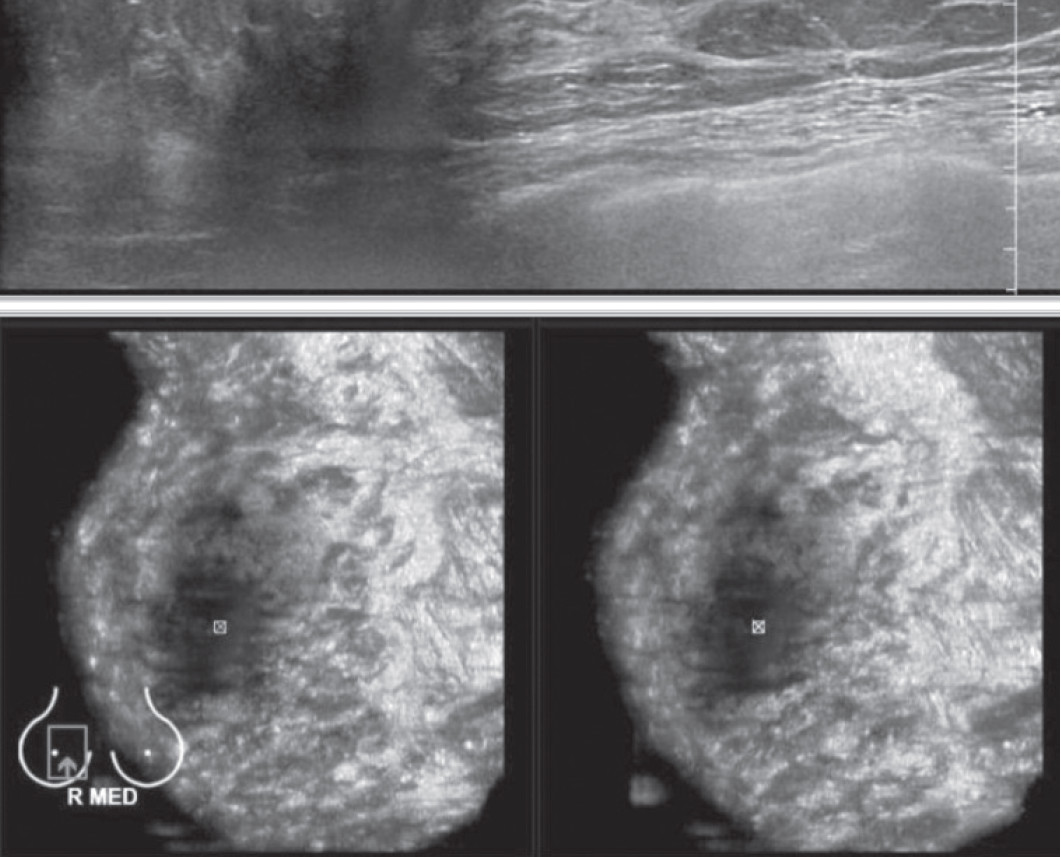
Figure 6: Shows nipple artifact causing limitation of lesion detected under the nipple.
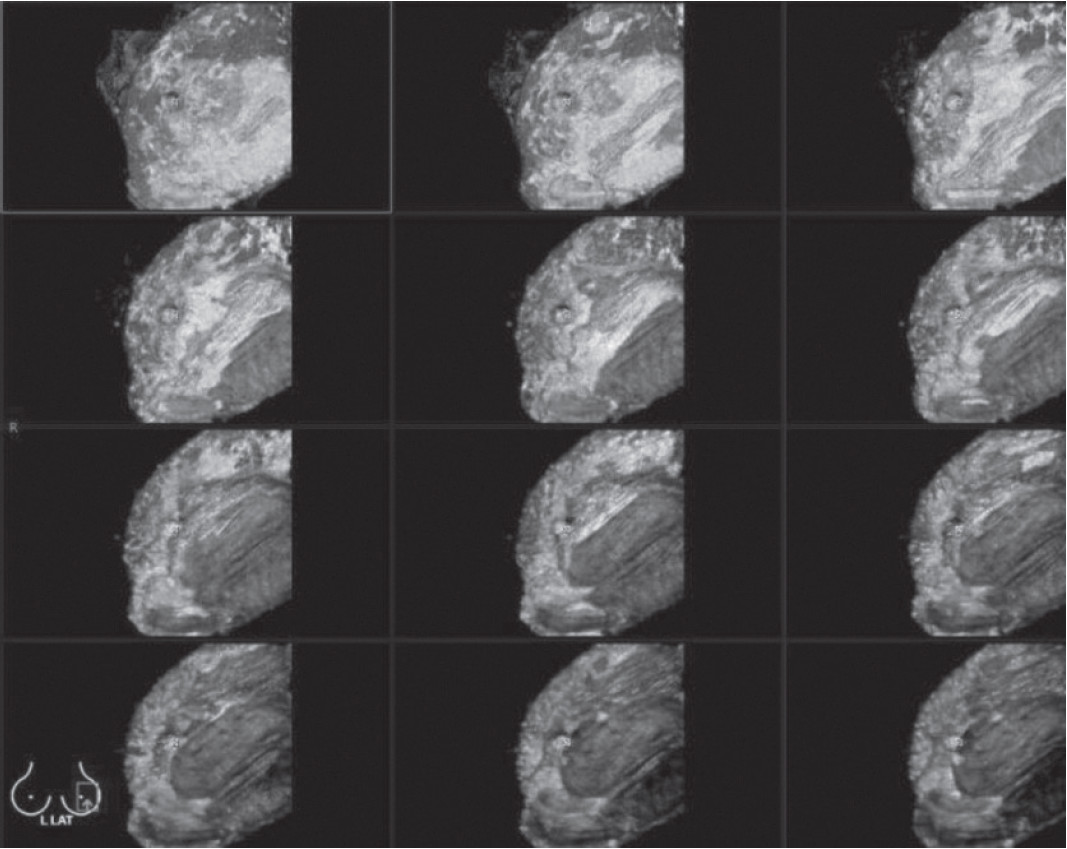
Figure 7: Shows motion artifact from patient movement during the volume scan.
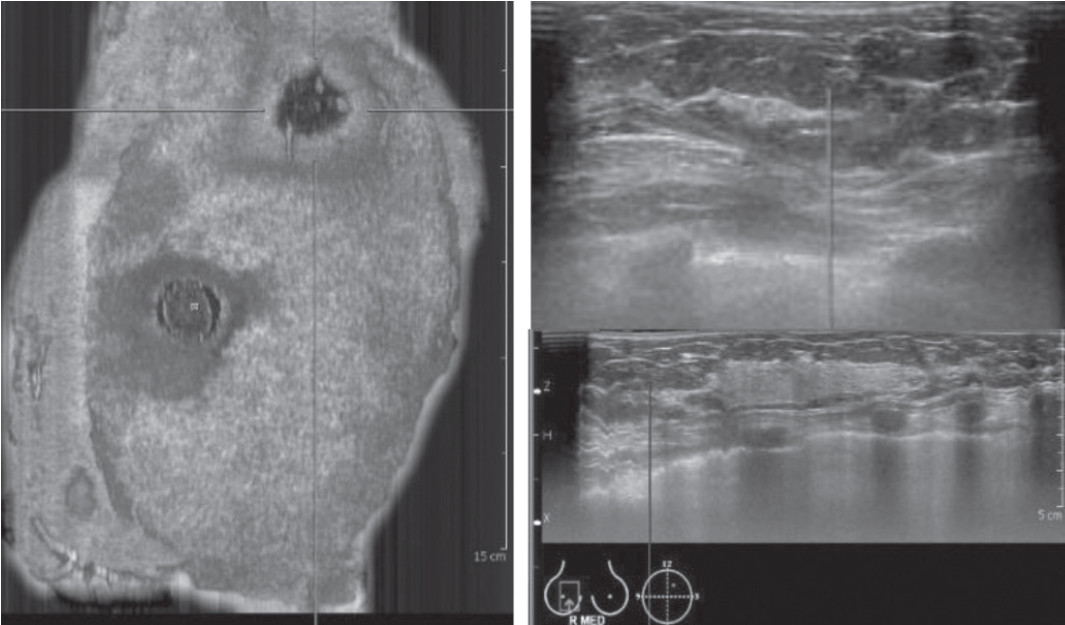
Figure 8: Shows contact artifact due to air trapped between skin and transducer.
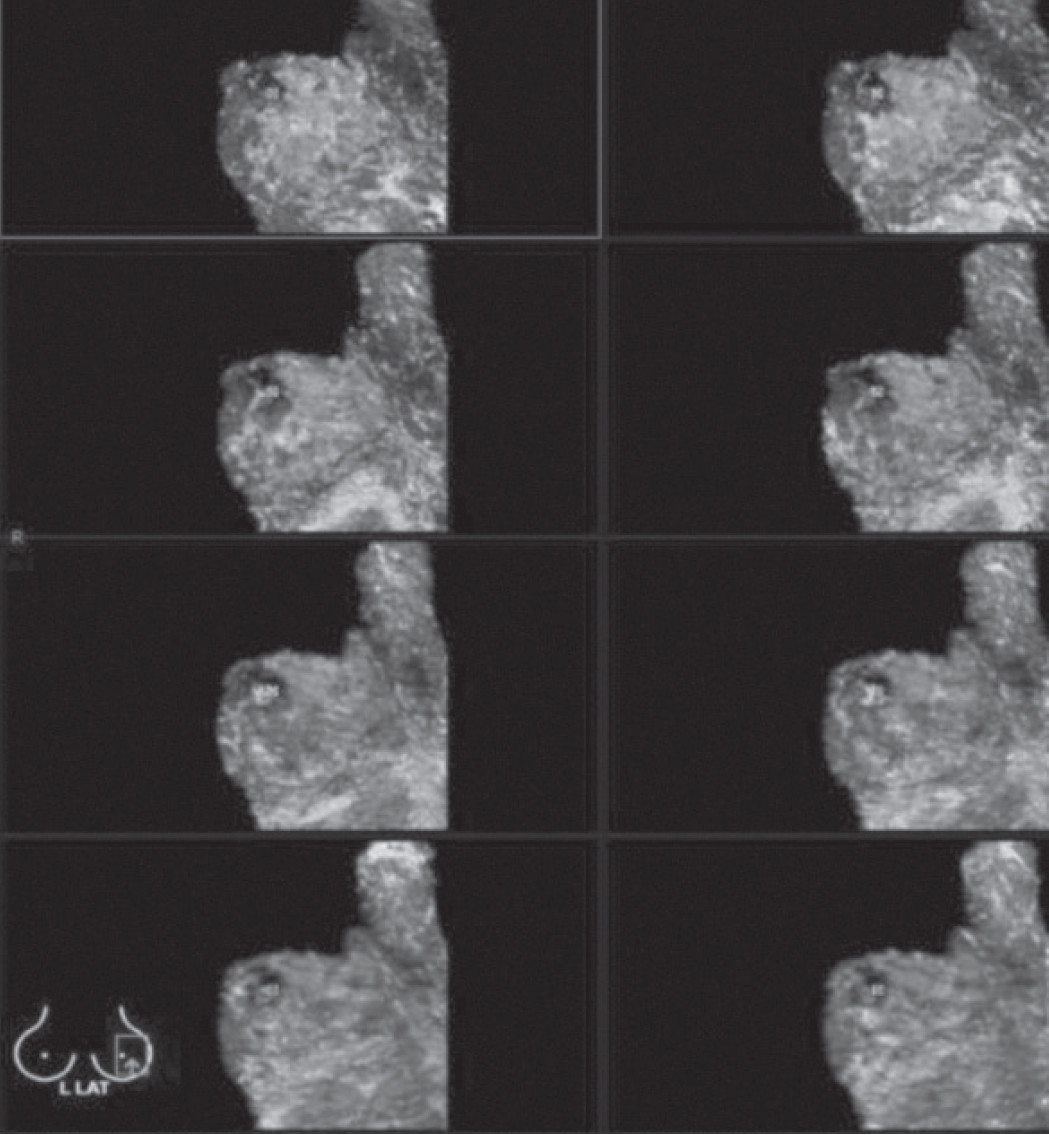
Figure 9: Shows out of view. The axillary adenopathy seen by mammography and HHUS is not included in the area scanned. Usually axillary lymph node is not totally inside the scanned area.
The result of the correlation of ABVS and HHUS findings were classified into 5 groups: both studies agreed in 306 out of 504 lesions (60.71%), both studies agreed but ABVS showed more lesions in 13 cases (2.58%). Both studies disagreed in 35 cases (6.94%), This was due to ABVS showing more detail and lesions were confirmed with scrolling for the fine details in 3 planes and 2 mm thickness. The echogenicity could be compared, for more confidence of differentiation of simple cysts from solid lesions. ABVS has no operator dependence, unlike HHUS which is known for its operator dependence.
ABVS was negative but HHUS was positive in 24 cases (4.76%), due to ABVS being able to confirm the continuity of fat lobule with subcutaneous fat by scrolling in 3 planes at 2 mm thickness, thus it could exclude the false positive lesions interpretation by HHUS. However, ABVS may not have included lesions in its FOV, due to our trying to limit the scan in order to reduce computer storage space, resulting in ABVS false negative.
HHUS was negative but ABVS was positive in 104 cases (20.63%). This was because HHUS has no multislice and multiplanar confirmation and is operator dependent, therefore, the lesions might not have been recognized, while all lesions in area scanned of ABVS were shown in details with confirmation.
From our study ABVS showed benefit over HHUS. Most of the different findings are due to the fact that ABVS has the benefits of a volume scan, whereby the images are readily displayed in multislice and multiplanar. When a lesion is suspected, a cursor is placed at that area and that particular point will be automatically displayed in other views with marker over that point. Scrolling over that point can be applied at 0.5-2.0 mm slice thickness, for better defining of the area, and confirmation or exclusion of the lesion.
ABVS has a large FOV, covers a large area of the breast, with nipple marking before the automatic volume scan which takes 1 minute for each scan, thus the lesion is standardized, reproducible and take less scanning time. ABVS is actually performed by the experienced technician. ABVS’s software adjusts measures and annotates the images on computer screen, and image quality is not different from that of CT volume scan.
However, ABVS had the drawbacks of nipple artifacts thus limiting. Technicians ought to look at the image which comes almost instantly after the scan. If artifacts are noted, technicians should then add a subareolar scan to the already available HHUS. Motion artifacts can be overcome by technicians should stopping, then repeating the scan after requesting the patient’s cooperation to remain motionless during the scan.
With contact artifacts, a repeat scan is recommended with more gel filling in the area to avoid air gapping. The technician should note this defect in the work sheet. The lesion may not be included in the scanned area, the out of view. We recommend technicians to check that the FOV includes the breast tissue to the most medial, lateral, superior and inferior parts of the breast, with overlapping scanned area. If mammography shows axillary adenopathy, the technician is urged to add axillary scan.
Overall benefits to patient with ABVS include it being a fast examination; breath holding is not required although the patient must stay completely still during the volume scan. There is no ionizing radiation which is perfect for young or pregnant patients. We did not use contrast agents, thus no related risk to the patient. Apart from that we used minimal compression, so it is more comfortable than mammography
ABVS has benefits to user as it is a volume scan, with standardized views, and reproducible. ABVS gives a streamlined workflow and the image in coronal plane is more familiar for surgeons and surgical planning, with more diagnostic information captured in less time. It captures volumes with online preview and offline review and manipulation, so it is beneficial in screening (mammography with US), using teleimaging. The technical limitations of HHUS are mostly overcome by ABVS.