Prostate cancer is the most commonly diagnosed cancer in menworldwide. Among men in the United State, prostate cancer isthe third leading cause of death from malignancy.1 According toAmerican Cancer Society, about 1 man in 7 will be diagnosed withprostate cancer during his lifetime.2 Prostate cancer develops mainlyin older men. About 6 cases in 10 are diagnosed in men aged 65 orolder, and it is rare before age 40. The average age at the time ofdiagnosis is about 66. Prostate cancer has high cure rate when detectedearly. Molecular Imaging technologies dramatically improve prostatecancer diagnosis and treatment. Although FDG is the most widely usePET tracer, using FDG for PET/CT imaging in prostate cancer islimited because a large fraction of prostate cancer shows limited FDGuptake.3 In the past decade, alternative tracers for prostate cancerimaging are radiolabeled choline derivative such as 18F-fluorocholineand 11C-choline. They are used as PET tracers for staging and restagingof prostate cancer.4 According to a meta-analyses report, in primarynodal imaging the specificity is as high as 95% but the sensitivity isvery poor (49%).5 Due to the lack of sensitive imaging for prostatecancer, more research has been focused on the development of newtracers that have better sensitivity and specificity. In recent years, targetingthe prostate specific membrane antigen (PSMA) with 68Ga-labeled and 18F-labeled PET tracers has gained highest clinical impact.
PSMA is a type II transmembrane glycoprotein that issignificantly (100-fold to 1000-fold) overexpressed in nearly allprostatic cancer cells compared with normal prostatic cells.6-9 Thelevel of PSMA expression rises with an increase in tumor grade,pathological stage and biochemical recurrence. PSMA PET/CT had asignificant impact on the management of prostate cancer. A cohort studyreported that radiotherapy management was changed for 50.8% whenusing 68Ga-PSMA-11 PET/CT for radiotherapy planning.10
Several imaging probes specifically targeting PSMA weredeveloped. Since the 1980s, several studies have been made totarget specific regions of the intracellular or extracellular domainof PSMA with monoclonal antibodies labeled with differentisotopes for nuclear medicine imaging.11 One of the first PSMAimaging agents was 111 In-labeled anti-PSMA antibody(111In-capromab pendetide, ProstaScint®).12 The effectiveness ofantibodies as diagnostic radiopharmaceuticals is limited by along circulating half-life resulting in a high unspecificbackground activity and poor tumor penetrability. Thus, theapplication of 111In-capromab pendetide for imaging of prostatecancer is limited because it has high non-specific uptake andrelatively poor tumor-to-background ratios.
Besides the development of PSMA monoclonalantibodies, small molecule PSMA inhibitors with high affinitygained a lot of interest. Because of their small size, they havebetter tumor penetration than antibodies. A series of studieshave been made to evaluate the role of small moleculeinhibitors of PSMA labeled with 123I, 99mTc, 18F, 111In, and68Ga.13-24 PSMA inhibitors fall into 3 families: urea-based,phosphorous-based, and thiol-based 25 as shown in Figure 1.A study by Chen et al., compared PSMA ligands with differentlinker lengths and has shown that an increased linker lengthenhanced the affinity for PSMA and increased tumor uptake.26Urea-based inhibitors have a high affinity and specificityfor PSMA and fast and efficient internalization into the cells.Several clinical studies evaluating PSMA ligands have beenperformed. Examples of small molecule PSMA ligands areshown in Figure 2. Among these agents, the 68Ga- and18F-labeled compounds have attracted the most attentionbecause they can be used for PET/CT imaging. Currently, themost widely used PET tracer for prostate cancer imaging is68Ga-PSMA-11.24 68Ga-PSMA-11 has many synonyms. Hereis the list of 68Ga-PSMA-11 in difference writing.
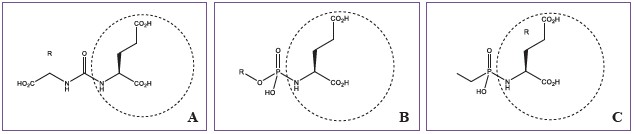
Figure 1: Small molecule PSMA inhibitors for prostate cancer studies (A) the urea-based compounds, (B) the glutamatephosphoramidates and (C) the 2-(phosphinylmethyl) pentanedioic acids. The binding domains are shown in the circle.
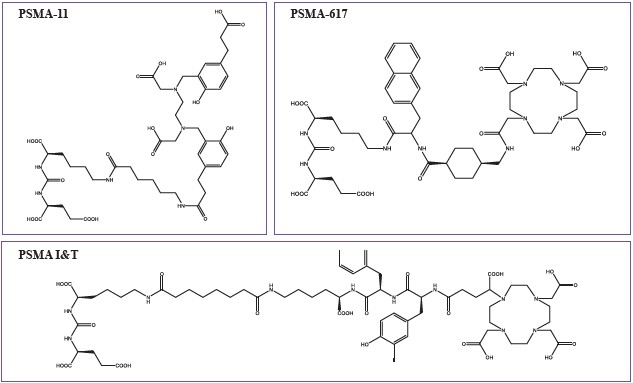
Figure 2: Example of small molecule PSMA ligands.
68Ga-PSMA-11 has a disadvantage with respect toproduction capacity and nuclear properties. The half-life of 68Ga is only 68 minutes. Therefore, delivery of sufficientamount of tracer activities to a remote center is quite challenging.68Ga (Gallium-68) is produced with 68Ge (Germanium-68)generator. Preparation of 68Ga-PSMA-11 is relatively easy.One batch of production of 68Ga-PSMA-11 can be used with 2-4 patients per generator elution. The main advantage of 68Ga-PSMA-11 is the commercially availability of 68Ge/68 Gagenerators. The long half-life of 68Ge (271 days) permits thegenerator to be used for several months or up to a year. Theshort half-life of 68Ga (68 minutes) allows multiple elution ofthe generator on the same day. For a center that does not haveaccess to a cyclotron and has a moderate number of patients,the price of these generators is a reasonable investment. 68Ge/68Ga generator was first developed in 1950. Thefirst commercial 68Ge/68Ga generator was introduced in late1990s which resulted in the blossoming of the 68Ga-PET.Pharmaceutical grade generators appeared on the market in 2014. Examples of commercial 68Ge/68Ga generator are shownin Figure 3. A generator is a self-shield system housing aparent/daughter radionuclide mixture in equilibrium. Figure 4 shows a schematic presentation of the cross section of acolumn-based generator. Commercial generators consist of ashort chromatographic column packed with a solid support ina shielding container. 68Ge which is produced from a highenergy cyclotron from stable 69Ge isotope is absorbed onto acolumn filled with inorganic, organic or mixed matrix. 68Gedecays to 68Ga and 68Ga decays to stable 68Zn as shown inFigure 5. 68Ga is washed off the column with an appropriatesolution. Then 68Ga can be used for tracers labeling.Figure 3: Example of commercial 68Ge/68Ga Generators.Figure 4: Schematic presentation of the cross section of acolumn-based generator.Figure 5:

Figure 3: Example of commercial 68Ge/68Ga Generators.
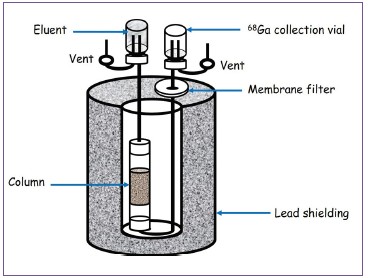
Figure 4: Schematic presentation of the cross section of acolumn-based generator.
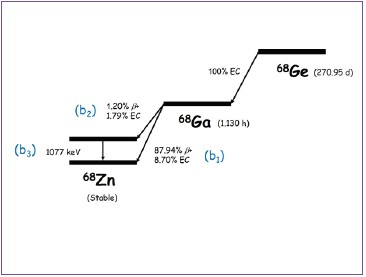
Figure 5: Schematic decay of 68Ge.
PSMA-11 contains the chelator HBED-CC (N,N’-bis[2-hydroxy-5-(carboxyethyl)benzyl] ethylenediamine –N,N’-diacetic acid). The chelator HBED-CC allows labelingwith kit formulations at room temperature without criticalradiochemistry demands.27 68Ga-PSMA-11 can be preparedby several methods. It can be prepared with an automatesynthesis system which can provide the reliability,reproducibility and safety of radiopharmaceutical productions.In recent times, the widespread, routine clinical use of 68Ga-PSMA-11 demands availability of a ready-to-use kitformulation to enable convenient radiopharmaceuticalpreparation. A freeze-dried kit vial for formulation of 68Ga-PSMA-11 was developed by a number of centers.28,29 Thismethod will provide convenient preparation of 68Ga-PSMA-11.Satpati D., et al reported that 68Ga-PSMA-11 could be preparedin > 98 % radiochemical yield and purity using the freeze-driedkit vials. The development of a simple and ready-to-usefreeze-dried kit for preparation of 68Ga-PSMA-11will contributeto a major step towards the widespread use of 68Ga-PSMA-11for prostate cancer imaging with PET/CT.
68Ga has physical half-life of only 68 minutes.Therefore, delivery of sufficient radiopharmaceutical activitiesto a remote center is challenging. One batch of 68Ga-PSMA-11production can be used with 2-4 patients. In large centers withmany patients, several productions per day are required, ormultiple generators are needed to produce sufficient amountof activities. The centers which have quantitative demand, theuse of 18F-labeled PSMA tracers may be more efficient. Highactivities of 18F can be produced with a cyclotron. Thephysical half-life of 18F is 109.77 minutes. The 18F-labeledPSMA tracers are developed. Two promising 18F-labeledPSMA tracers which are under clinical investigation are18F-DCFPyL and 18F-PSMA-1007. PSMA-1007 is((3S,10S,14S)-1-(4-(((S)-4-carboxy-2-((S)-4-carboxy-2-(6-18F-fluoronicotinamido)butanamido)butanamido)methyl)phenyl)-3-(naphthalen-2-ylmethyl)-1,4,12-trioxo-2,5,11,13-tetraazahexadecane-10,14,16-tricarboxylic acid) and DCFPyLis (2-(3-{1-carboxy-5-[(6-18F-fluoro-pyridine-3-carbonyl)-amino]-pentyl}-ureido)-pentanedioic acid). The chemicalstructures of 18F-PSMA-1007 and 18F-DCFPyL are shown inFigure 6. Due to longer half-life of 18F and the possibility toproduce in high activity, the 18F-labeled PSMA enable centralizedproduction and delivery to distant centers. 18F has a lowerpositron energy than 68Ga (0.68 MeV for 18F vs. 1.90 MeV for68Ga). Thus PET imaging with 18F-labeled tracers have betterspatial resolution than 68Ga-labeled tracers.
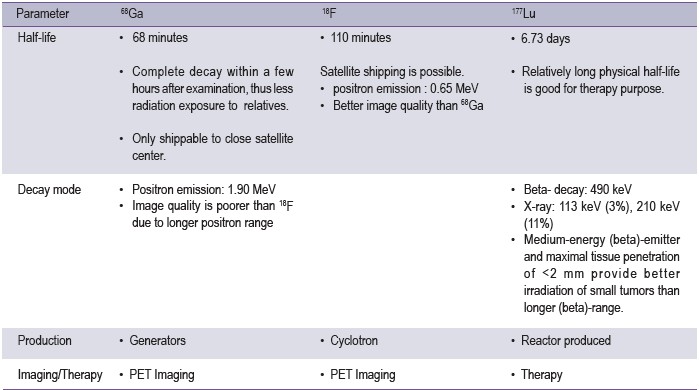
Besides diagnostic imaging, radiolabeled PSMA ligandsalso have potential for radionuclide therapy of prostate cancer.Several PSMA ligands are currently investigated clinically fordiagnostic and therapeutic purposes. Some PSMA ligands canbe labeled with either 68Ga for PET imaging or 177Lu(Lutetium-177) for radionuclide therapy. 177Lu physicalproperties are good to use as therapeutic radionuclide. 177Lu isa medium-energy beta emitter (490 keV) with a maximumtissue penetration of < 2 mm. The emission characteristicsmatch the lesion size/volume to be treated to ideally focusenergy within the tumor rather than in the tissue surroundingthe lesion. 177Lu has a relatively long physical half-life of 6.73days. These physical properties of 177Lu allow for the deliveryof a high radiation dose to prostate cancer cells. These PSMAligands which can be labeled either with 68Ga or 177Lu havepotential to be used for both diagnostic and therapeuticpurposes. Example of such agents are PSMA I&T andDKFZ-617. PSMA I&T (for Imaging and Therapy) isDOTAGA-(I-y)fk(Sub-KuE). It is DOTAGA-couple PSMAligands by increasing the hydrophilicity of the ligand bysubstitute DOTA by 1,4,7,10-tetraazacyclodocecane-,1-(glutaric acid)-4,7,10-triacetic, resulting in DOTAGAFFK(Sub-KuE) which can be labeled with both 68Ga and177Lu.30 DOTA-conjugated DKFZ-617 PSMA ligand isanother tracer which has mainly been used for therapy may beused for diagnostic application too. The results show that177Lu-PSMA is a safe treatment option for metastatic prostatecancer patients and has a low toxicity profile.Positive responses to therapy in terms of decline in PSAare reported and more than 40% of patients showed more than50 % PSA decline.31,32 Comparison of the properties of 68Ga,18F, and 177Lu is shown in Table 1.
Positive responses to therapy in terms of decline in PSAare reported and more than 40% of patients showed more than50 % PSA decline.31,32 Comparison of the properties of 68Ga,18F, and 177Lu is shown in Table 1.
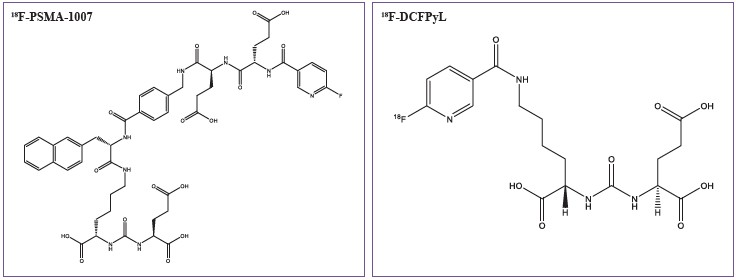
Figure 6: 18F-PSMA-1007 and 18F-DCFPyL.
Table 1: Comparison of 68Ga, 18F and 177Lu
It appears that PSMA shows great promise not just in detecting prostate cancer, but also as a target for radionuclide therapy. At present, there are many radiolabeled PSMA ligands available for imaging and therapy. 68Ga-PSMA-11 is currently the most widely used for prostate cancer imaging with PET/CT. Development of 18F-PSMA shows promising results. In the future, PSMA imaging will be used more widely due to the availability of tracers. The use of PSMA PET/CT resulted in a change of the therapeutic management in up to 50% of cases. The currently available data clearly shows that PSMA imaging has a clinical impact on the diagnosis of prostate cancer. Radiolabeled PSMA tracers also have high potential for therapeutic approaches.
The author thanks the PET center and Cyclotron facility team at Wattanosoth Cancer Center including doctors, nurses, physicists, technologists, chemists, engineers and pharmacists. During the past 12 years, they have put a lot of effort into continued improvements in quality and range of services. Also, the author is very grateful for the support from the executive of the Bangkok Hospital and Wattanosoth Cancer Hospital.