Catheter-related bloodstream infections (CRBSIs) are associated with substantial morbidity and cost. In the United States alone, over 250,000 cases occur each year with a mortality rate as high as 25%, increasing both the length of hospital stay and risks for other diseases like endocarditis and osteomyelitis.1 In addition to the direct serious health consequences, CRBSIs also come at a heavy financial price, costing the healthcare system an estimated 2.68 billion USD annually.2-5 Central line maintenance bundles–systems of interventions that often include hand-hygiene training, maximal sterile barrier precautions during central line insertion, use of chlorhexidine during skin preparation, and avoidance of the femoral placements for central lines–have generated a lot of excitement for their effectiveness at reducing rates of CRBSIs in intensive care unit (ICU) settings.6-9 However, compliance with the established protocols remains a key challenge with some studies reporting healthcare professionals following the recommendations only 35-45% of the time.10,11
Consequently, there has been a growing imperative to investigate novel and more passive technologies, which can improve prevention rates for CRBSIs while reducing the workload placed on hospital staff. Testing these new technologies aimed at preventing CRBSIs will first require a sensitive and reproducible method of introducing appropriate contaminants into in vitro systems in order to accurately evaluate any antiseptic effects. Historically, the preferred inoculation model has been the dip method, where devices of interest such as catheter hubs or needleless connectors are immersed in a suspension of bacteria for a period of time and then allowed to dry.12-16 However, significant variability between studies in the specifics of their methods, including differences in solutions, concentrations, immersion time and drying time, make it difficult to compare their effectiveness. Moreover, many of the studies fail to quantify the bacteria inoculated, instead reporting solely the presence or absence of bacteria on inoculated surfaces after disinfection, ignoring the possibility of irregular applications.12,14,15,17
Therefore, this study aimed to test results from the conventional dip method against a novel “Inoculation cap” or “Inocap” model. The Inocap model proposed in this study was designed to have better control of the target area for inoculation around the outer surfaces of the luer hub, transfer relevant and consistent amount of bacteria that occur inside hospitals, and increase repeatability between samples by reducing human error. This model would be representative of the contamination process commonly occurring in hospital settings, leading to intraluminal infection in catheterized patients. Methicillin-resistant Staphylococcus aureus (MRSA) was chosen for this study due to its character as one of the most commonly found pathogens associated with nosocomial infections.18
Bacteria Stock Suspension Preparation in TSB and NSS
Methicillin-resistant Staphylococcus aureus (MRSA) ATCC 43300 was used as model organism because MRSA is frequently responsible for CRBSIs and represents a highly antibiotic-resistant organism. MRSA ATCC 43300 colonies were suspended and streaked onto blood agar (Biomedia, Thailand) and incubated at 37°C for 48 hours by National Healthcare Systems Co., Ltd. (Thailand). The colonies were then diluted to the desired bacteria stock concentration, 0.5 McFarland Standard or ~1.5×108 colony-forming units (CFU)/ mL, in Tryptic Soya Broth (TSB, Himedia, India) or 0.9% Normal Saline Solution (NSS) depending of the experimental groups. Bacteria stock concentrations were measured using McFarland standards (DensiCHEK, Biomerieux, France) and further confirmed by serial dilution and spread on Tryptic Soy Agar (TSA).
Hub Inoculation
For the Inocap method, propylene vented male luer caps, no stem (Inoculation Cap Model No. 12084, Qosl stock diluents were tested with the Inocap method for relevant and consistent of quantity of bacteria inoculated. There was a significant difference (p < 0.0001) in the average bacteria counts inoculated between the NSS and TSB bacterial stock diluents. The TSB solution yielded 3.60×105 CFU/mL, which is a 2.5-log reduction from the 1.25×108 CFU/ mL of stock concentration. The NSS solution inoculated lower amount of bacteria on to the female hub. It yielded 1.14×104 CFU/mL, which is a 4.0-log reduction from the 1.14×108 CFU/ mL stock concentration (Figure 3b). There was also a significant difference in the variance between the two groups (p < 0.0001), indicating that polymer surfaces found (Figure 3a), with those subjected to the Inocap method showing a smaller standard error (Table 1). Moreover, there was no significant difference in the means of each group (p > 0.25), suggesting that the Inocap method was able to produce the same desired final inoculated concentration of approximately ~1.5×105 CFU/mL with greater reliability.
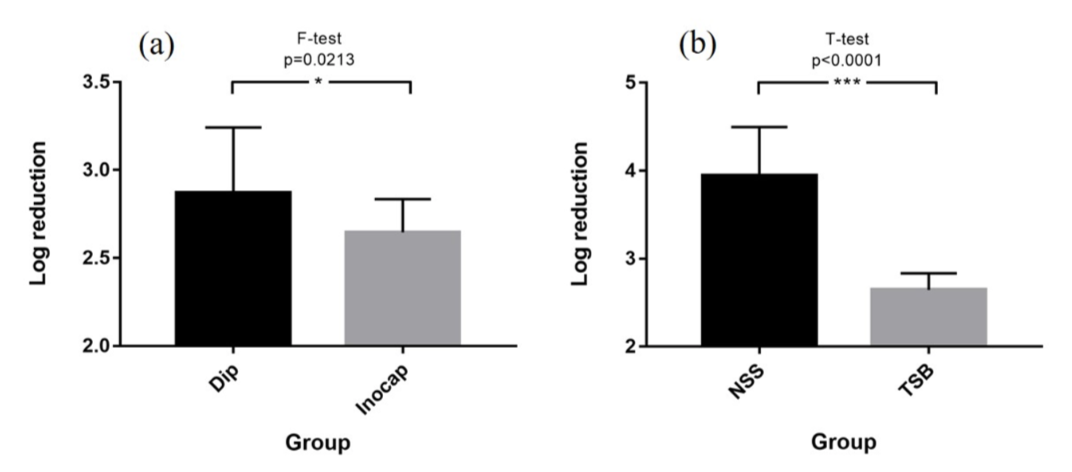
Figure 3: Log reduction showing the amount of inoculated CFU harvested from luers relative to the initial starting bacteria stock concentration. (a) Inocap and Dip methods show no significant difference in the average amount inoculated, but the dip method yields a higher variance. (b) All luers for both the TSB and NSS groups were inoculated using the Inocap method. F-test shows significant difference in variance between TSB and NSS and the TSB group showed significantly less log reduction than the NSS group.
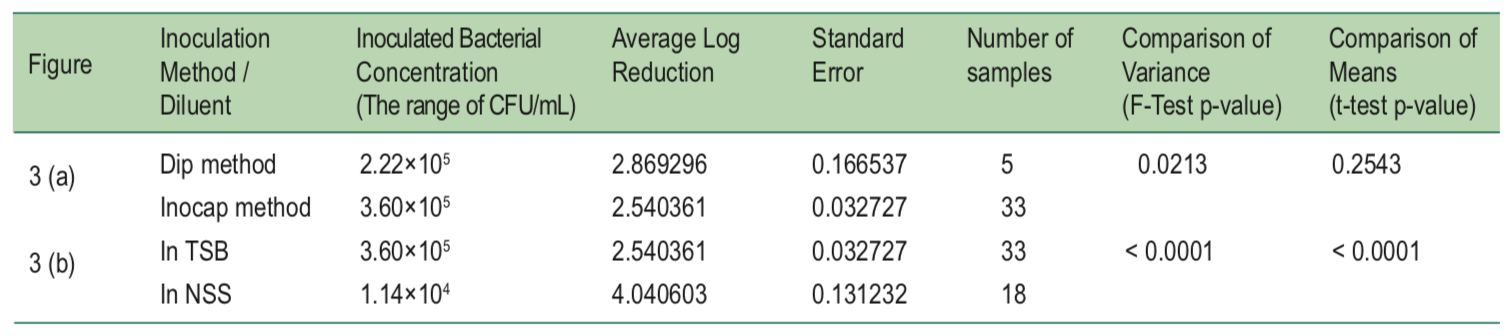
Table 1: A summary of the standard error, average log reduction measurements, and F-test and t-test p-values for the different groups of luers organized by inoculation method and inoculum diluent
Bacteria Suspensions Using TSB Inoculate Higher Concentrations for Aseptic Tests
Previous literature used various bacterial stock diluents to prepare bacteria inoculation solution.12-14 In this study, two different solutions, TSB and NSS, were used as bacterial stock diluents. The bacterial stock diluents were tested with the Inocap method for relevant and consistent of quantity of bacteria inoculated. There was a significant difference (p < 0.0001) in the average bacteria counts inoculated between the NSS and TSB bacterial stock diluents. The TSB solution yielded 3.60×105 CFU/mL, which is a 2.5-log reduction from the 1.25×108 CFU/ mL of stock concentration. The NSS solution inoculated lower amount of bacteria on to the female hub. It yielded 1.14×104 CFU/mL, which is a 4.0-log reduction from the 1.14×108 CFU/ mL stock concentration (Figure 3b). There was also a significant difference in the variance between the two groups (p < 0.0001), indicating that polymer surfaces found (Figure 3a), with those subjected to the Inocap method showing a smaller standard error (Table 1). Moreover, there was no significant difference in the means of each group (p > 0.25), suggesting that the Inocap method was able to produce the same desired final inoculated concentration of approximately ~1.5×105 CFU/mL with greater reliability. inoculated using TSB with the Inocap method yielded higher concentrations of bacteria and smaller variations, than those prepared with NSS.
The results were presented as a log reduction in bacteria stock concentration to normalize for experimental variation in the starting stock concentrations between the groups. The data showed that the inocap method had no significant difference in average inoculated bacterial count as the dip method but had a lower variance (Figure 3a). This means that the inocap model can reduce uncontrollable model error in experiments testing for disinfectant efficacy studies. It is important to acknowledge that the greater variance seen with the dip method could in part be due to the smaller sample size of the dip group (5 compared to 33), but this was taken into consideration when performing statistical analysis. The improved consistency of the inocap method can be explained by how it standardizes the inoculation procedure in two ways, first by mixing and then promptly aliquoting the stock solution to ensure equal distribution, and secondly by using an O-ring and threads along the luer connector to control the surface that comes into contact with the luer. This is in contrast with the dip method that requires scientists to manually submerge each luer sequentially in the stock solution, with no control over the concentration of bacteria currently in contact with the surface, nor the exact surface area that comes into contact with the stock solution.
The reason why bacteria stock solutions prepared in NSS inoculated much lower concentrations onto the luer connectors (Figure 3b) could be due to the viscosity of the solution or the fact that TSB is a buffered enrichment medium with proteins and nutrients, and can support bacterial activity. NSS lacks the ingredients that support bacterial activity but is still able to prevent lysis of bacterial cells. Viable bacterial cells remained suspended in NSS, and the majority is removed from the Inocaps with the removal of the inoculum fluid.
Converting from analytical solution concentration of inoculated female luer (CFU/mL) or TSB bacteria stock concentration to absolute counts (CFU), the average bacteria count on luers using NSS-Inocap method was 1.14×105 CFU, whereas the average bacteria count on luers using TSB-Inocap method was 3.60×106 CFU. The TSB-Inocap method counts were closer to the desired value of 1.0×106 CFU and could be fine-tuned further by adjusting the initial inoculum concentration. Bacterial count of 1.0 x 106 CFU is desired because it is sufficiently high to prove a 4-log reduction efficacy for medical device disinfection as required by the United States Food and Drug Administration (FDA) and the within the upper bound of the bacteria count commonly found in hands of medical personnel.20 According to the Centers for Disease Control and Prevention (CDC) transient microflora on hands of healthcare workers from hand contact is a common source of contamination in hospital settings.20 Consequently, the use of TSB as the diluent was found to be more suitable for the Inocap method since it achieved quantities within the desired range explained above. The higher variance from NSS group indicated that bacterial stock diluent is also one of the factors that strongly affect the consistency of the inoculation concentration.
This study developed and investigated a new “Inocap” method for standardized bacteria inoculation on to controlled target surfaces of catheter hubs. The results showed a significantly smaller variance in the number of bacteria inoculated onto the catheter hubs by the new Inocap method than the traditional dip method (p ≈ 0.02) while producing no significant difference in the mean value (p > 0.25), indicating that this new Inocap method could work as a more precise inoculation model for future disinfection studies.
TSB and NSS were also tested as the bacterial stock diluents with the Inocap method and found a significant difference in the average amount of inoculated bacteria, with TSB solutions yielding higher amount inoculated than NSS solutions. A significant reduction in the variance was also found, indicating that the use of TSB as the diluent with the Inocap method provided a better consistency during the inoculation process.
This work was supported by Bangkok Dusit Medical Services. The authors would like to thank Chiyos Kunanusont, M.D., PhD., Pravich Tanyasittisunthorn, M.D., Rita Juneja, PhD., Panisa Sundravorakul, Aonnicha Burapachaisri, and the members of CassLab for their comments and support with this project and manuscript. We thank Chirotchana Suchato, M.D. and Shanta Modak, PhD. for their guidance.